Stainless Steel - High Temperature Resistance
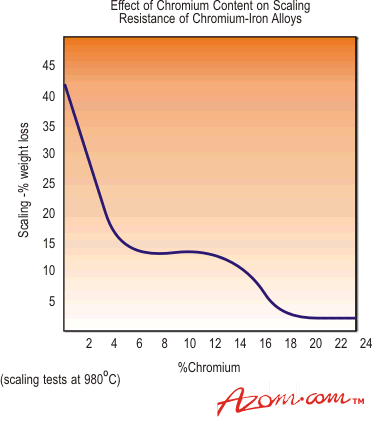
Stainless steels are most commonly used for their corrosion resistance. The second most common reason stainless steels are used is for their high temperature properties; stainless steels can be found in applications where high temperature oxidation resistance is necessary, and in other applications where high temperature strength is required. The high chromium content which is so beneficial to the wet corrosion resistance of stainless steels is also highly beneficial to their high temperature strength and resistance to scaling at elevated temperatures, as shown in the graph of Figure 1. | |||||||||||||||||||||||||||||||||
Scaling Resistance | |||||||||||||||||||||||||||||||||
Resistance to oxidation, or scaling, is dependent on the chromium content in the same way as the corrosion resistance is, as shown in the graph below. Most austenitic steels, with chromium contents of at least 18%, can be used at temperatures up to 870°C and Grades 309, 310 and 2111HTR (UNS S30815) even higher. Most martensitic and ferritic steels have lower resistance to oxidation and hence lower useful operating temperatures. An exception to this is the ferritic grade 446 - this has approximately 24% chromium, and can be used to resist scaling at temperatures up to 1100°C.
Figure 1. Effect of chromium content on scaling resistance Table 1 shows the approximate maximum service temperatures at which the various grades of stainless steels can be used to resist oxidation in dry air. Note that these temperatures depend very much on the actual environmental conditions, and in some instances, substantially lower temperatures will result in destructive scaling. Table 1. Maximum service temperatures in dry air, based on scaling resistance (ref: ASM Metals Handbook)
| |||||||||||||||||||||||||||||||||
Creep Strength | |||||||||||||||||||||||||||||||||
The high-temperature strength of materials is generally expressed in terms of their "creep strength" - the ability of the material to resist distortion over long term exposure to a high temperature. In this regard, the austenitic stainless steels are particularly good. Design codes such as Australian Standard AS1210 "Pressure Vessels" and AS4041 "Pressure Piping" (and corresponding codes from ASME and other bodies) also stipulate allowable working stresses of each grade at a range of temperatures. The low carbon versions of the standard austenitic grades (Grades 304L and 316L) have reduced strength at high temperature so are not generally used for structural applications at elevated temperatures. "H" versions of each grade (eg 304H) have higher carbon contents for these applications, which results in significantly higher creep strengths. "H" grades are specified for some elevated temperature applications. Although the duplex stainless steels have good oxidation resistance due to their high chromium contents, they suffer from embrittlement if exposed to temperatures above about 350°C, so they are restricted to applications below this. Both martensitic and precipitation hardening families of stainless steels have high strengths achieved by thermal treatments; exposure of these grades at temperatures exceeding their heat treatment temperatures will result in permanent softening, so again these grades are seldom used at elevated temperatures. Structural StabilityThe problem of grain boundary carbide precipitation was discussed under intergranular corrosion. This same phenomenon occurs when some stainless steels are exposed in service to temperatures of 425 to 815°C, resulting in a reduction of corrosion resistance which may be significant. If this problem is to be avoided the use of stabilized grades such as Grade 321 or low carbon "L" grades should be considered. A further problem that some stainless steels have in high-temperature applications is the formation of sigma phase. The formation of sigma phase in austenitic steels is dependent on both time and temperature and is different for each type of steel. In general Grade 304 stainless steel is practically immune to sigma phase formation, but not so those grades with higher chromium contents (Grade 310) with molybdenum (Grades 316 and 317) or with higher silicon contents (Grade 314). These grades are all prone to sigma phase formation if exposed for long periods to a temperature of about 590 to 870°C. Sigma phase embrittlement refers to the formation of a precipitate in the steel microstructure over a long period of time within this particular temperature range. The effect of the formation of this phase is to make the steel extremely brittle and failure can occur because of brittle fracture. Once the steel has become embrittled with sigma it is possible to reclaim it by heating the steel to a temperature above the sigma formation temperature range, however, this is not always practical. Because sigma phase embrittlement is a serious problem with the high silicon grade 314, this is now unpopular and largely replaced by high nickel alloys or by stainless steels resistant to sigma phase embrittlement, particularly 2111HTR (UNS S30815). Grade 310 is also fairly susceptible to sigma phase formation in the temperature range 590 to 870°C, so this "heat resistant" grade may not be suitable for exposure at this comparatively low-temperature range and Grade 321 is often a better choice. Environmental FactorsOther factors that can be important in the use of steels for high-temperature applications are carburization and sulfidation resistance. Sulfur bearing gases under reducing conditions greatly accelerate the attack on stainless alloys with high nickel contents. In some instances Grade 310 has given reasonable service, in other grade S30815, with a lower nickel content is better, but in others a totally nickel-free alloy is superior. If sulfur-bearing gases are present under reducing conditions it is suggested that pilot test specimens be first run under similar conditions to determine the best alloy. Thermal ExpansionA further property that can be relevant in high-temperature applications is the thermal expansion of the particular material. The coefficient of thermal expansion is expressed in units of proportional change of length for each degree increase in temperature, usually x10-6/°C, μm/m/°C, or x10-6cm/cm/°C, all of which are identical units. The increase in length (or diameter, thickness, etc) can be readily calculated by multiplying the original dimension by the temperature change by the coefficient of thermal expansion. For example, if a three-meter long Grade 304 bar (coefficient of expansion 17.2 μm/m/°C) is heated from 20°C to 200°C, the length increases by: 3.00 x 180 x 17.2 = 9288 μm = 9.3 mm The coefficient of thermal expansion of the austenitic stainless steel is higher than for most other grades of steel, as shown in the following table. Table 2. Coefficient of thermal expansion - average values over 1-100°C This expansion coefficient not only varies between steel grades, it also increases slightly with temperature. Grade 304 has a coefficient of 17.2 x 10-6/°C over the temperature range 0 to 100°C but increases above this temperature The effect of thermal expansion is most noticeable where components are restrained, as the expansion results in buckling and bending. A problem can also arise if two dissimilar metals are fabricated together and then heated; dissimilar coefficients will again result in buckling or bending. In general, the quite high thermal expansion rates of the austenitic stainless steels mean that fabrications in these alloys may have more dimensional problems than similar fabrications in carbon or low alloy steels, in ferritic, martensitic or duplex stainless steels. The non-austenitic stainless steels also have somewhat higher thermal conductivities than the austenitic grades, which may be an advantage in certain applications. Localized stresses from expansion during heating and cooling can contribute to stress corrosion cracking in an environment that would not normally attack the metal. These applications require the design to minimize the adverse effects of temperature differentials such as the use of expansion joints to permit movement without distortion and the avoidance of notches and abrupt changes of section. |
This Article is from AZoM.com